Blog
-

“Hear With Light”… Earlens Raises $51M to Launch Light-driven Hearing Aid
 21 Jun , 2016
After receiving approval from the Food and Drug Administration (FDA) late last year, hearing device maker Earlens raised $51 million in debt and equity to back the launch of its light-driven hearing aid. The FDA cleared it via its de novo premarket review pathway last September; that route is reserved for low- to moderate-risk medical devices that do not have a substantially equivalent marketed device.
21 Jun , 2016
After receiving approval from the Food and Drug Administration (FDA) late last year, hearing device maker Earlens raised $51 million in debt and equity to back the launch of its light-driven hearing aid. The FDA cleared it via its de novo premarket review pathway last September; that route is reserved for low- to moderate-risk medical devices that do not have a substantially equivalent marketed device.According to the website FierceMedicalDevices, which covers the medical devices and diagnostics industries, hearing aids are challenging to market. Earlens is reportedly aiming to offer its novel technology at a cost that may be slightly higher than existing options.
“We intend to execute a limited launch of our technology as we refine our commercial approach. Once we have built experience in the market, we will expand our launch footprint, eventually beginning direct to patient marketing activities in support of our trained clinics,” EarLens CTO Brent Edwards told FierceMedicalDevices.
He continued, “Given the performance of our technology, we will be pricing our product at a premium to conventional hearing aid technologies.”
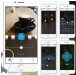
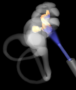
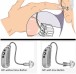
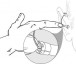
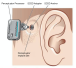
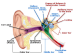
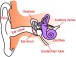
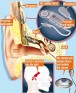
The Earlens Contact Hearing Device (CHD) includes both a tympanic membrane transducer, which is placed deeply into the ear canal on the eardrum without surgery, and a behind-the-ear audio processor that sits on the outer ear and is connected to an ear tip that is placed in the ear canal.
Sound waves are converted by the system to electronic signals, which are digitally processed, amplified and sent to the ear tip. A laser diode converts the signals into pulses of light, which shine onto a photodetector in the tympanic membrane transducer. This then converts the light back into electronic signals to transmit sound vibrations directly to the eardrum.
In a 48-subject trial over 30 days, Earlens offered a 33% average improvement in word recognition. Users also gained function in higher frequencies, which is typically not found with conventional air-conduction hearing aids.
For more details, see the full report on theFierceMedicalDevices website.
Source: Earlens, FierceMedical
Devices Image credit: Earlens; FierceMedicalDevices