Blog
-
Eosera to Introduce “EarwaxMD” at AudiologyNOW! 2017.
 6 Apr , 2017
6 Apr , 2017
Eosera to Introduce “EarwaxMD” at AudiologyNOW! 2017.
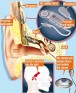
EOSERA™, Inc, Fort Worth, Tex, announced the launch of a new earwax impaction solution, Earwax MD™, to hearing aid centers and audiologists at the AudiologyNOW! 2017Conference in Indianapolis, Indiana from April 5 to 8, 2017. EOSERA states that it is the healthcare industry’s first earwax impaction solution to safely and effectively dissolve wax blockage in a single dosage.
People who use the hashtag#EarwaxMDAAA on social media, will be entered in a drawing to win an Apple Watch. Post on social media using the hashtag #EarwaxMDAAA to enter a drawing to win one of the watches. From now until the conference ends, Eosera will put names in a drawing for every social media post with its hashtag.
“We are thrilled that the AudiologyNOW! conference coincides with our April launch,” said Elyse Dickerson, founder and CEO of Eosera Inc. “We know that Earwax MD will help people worldwide with earwax impaction, especially those who are more prone to the condition such as the elderly and hearing aid users. By bringing this novel innovation to market, our goal is to improve the lives of both doctors and patients by saving people time and money. Patients can now proactively clean their ears at home which means less visits to the doctor for earwax removal. Instead of having to dig out earwax, audiologists and hearing aid centers can now fit patients for hearing aids in one visit.”
Dickerson advises patients to ask their local hearing aid centers about the product.
“We look forward to educating audiologists and hearing specialists from companies like Beltone and Connect Hearing, and more, at the conference,” Dickerson said.
Clinical evaluations from Eosera.
According to Eoser, in a clinical evaluation of Earwax MD, a total of 30 ears with moderate to severe earwax impaction were tested. Eosera reports that a single application of Earwax MD (consisting of up to two doses) not only fully cleared the impaction in 86% of test subjects, but also reduced symptoms by 82%. Common symptoms of earwax impaction include a feeling of fullness, itching, ringing of the ears, and hearing loss.
In a separate study, researchers compared Earwax MD to two other competitive systems. According to Eosera, researchers collected samples of human earwax, placed them in test tubes with each solution, and measured the solutions’ effectiveness at dissolving the wax. Using a scale of 0 (no disintegration) to 4 (complete disintegration), wax samples were evaluated and scored at 5, 10, 15, and 30 minutes. At the end of 30 minutes, Earwax MD reportedly showed significant disintegration of the wax samples (scored 3.24), while Murine Ear and Debrox barely showed any change in the wax samples (scored 0.12).
Safe for human skin. Eosera also worked with a dermatologist to ensure Earwax MD was safe on skin. Fifty (50) subjects were exposed to Earwax MD in a series of tests designed to evaluate product safety. Skin tests showed no evidence of irritation.
Source: Eosera
Image credit: Eosera