Blog
-
Protein Movement in Inner Ear Hair Cells Shows Renewal Mechanism
 12 Nov , 2015
12 Nov , 2015
Scientists conducting zebrafish studies at Case Western Reserve University School of Medicine have announced the discovery that the movement of protein within hair cells of the inner ear shows signs of a repair and renewal mechanism.
The findings are highlighted in an article in the November 17, 2015 edition of Cell Reports. The authors say their discovery paves the way for one day therapeutically manipulating the movement of these proteins in a way that could reverse human hearing loss, (which affects approximately 15% of Americans between the ages of 20 and 69).
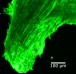
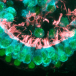
As the authors explain in the article, hearing is made possible when hair bundles protruding from the tops of hair cells capture the energy of sound waves, converting them into electrical signals that stimulate the auditory nerve to the brain. These hair bundles are made up of individual hair-like projections, or stereocilia, which sway in unison with other stereocilia. Extremely loud noise can force whipsaw motion of the stereocilia, causing them to be damaged, resulting in permanent hearing loss. Until now, the prevailing theory has been that the hair bundles, which last a lifetime, were made up of cellular scaffolding proteins that do not change or circulate.
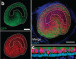
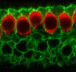
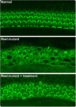
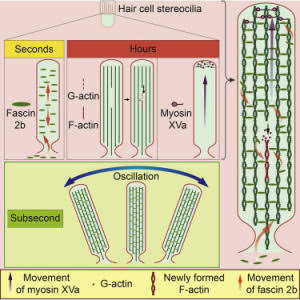
In their research, the investigators discovered that proteins in the hair bundle move at rapid rates, and this movement could explain why hair bundles last a lifetime. The researchers report that they used a confocal microscope to examine the movement of these proteins in the inner ears of baby zebrafish. The zebrafish at this early state of development are completely transparent, making it possible to view internal organs, including the ear structure, without dissection.
“What was surprising in our research with zebrafish is that proteins move so rapidly, implying that protein movement may be required to maintain the integrity of hair bundles in the inner ear,” said senior author Brian McDermott, PhD, associate professor of Otolaryngology-Head and Neck Surgery at Case Western Reserve University School of Medicine. “Our research tells us that constant movement, replacement and adjustment among proteins in the inner ear’s hair bundles serve a maintenance and even repair function.”
The research team discovered that vital proteins, actin and myosin, move at a leisurely pace over the course of hours. On the other hand, fascin 2b moves incredibly fast, in seconds. This discovery proved that the internal structure of stereocilia is dynamic, and the proteins move frequently rather than being mostly stationary. Further, the speed with which fascin 2b moves may facilitate repair of breaks in the actin cables of the hair bundle when the ear is exposed to loud noise.
When sound enters the ear, it causes intense vibrations and swaying movement of the hair bundle at extremely high rates. The research team found that what appears to make the hair bundle so durable in the face of such forces is the rate of protein turnover, which enables renewal of the hair bundle.
“It was once thought that most everything within the stereocilia was relatively immobile and static,” McDermott said. “We found that the constant, dynamic movement likely contributes to the permanency of the hair bundle structure to last a lifetime, or 70 to 90 years in human terms.”
McDermott’s team next plans to determine how the hair bundle may repair itself with such protein movement. They hypothesize that the process likely involves fascin 2b, but may also involve other proteins. “No one has shown that stereocilia heal themselves, so next we will find out if there is a repair mechanism,” McDermott said.
“People go deaf from damage to stereocilia in the inner ear. If we figure out ways of manipulating protein dynamics within stereocilia to heal this damage, we would likely be able to diminish hearing loss in humans.” Concluded McDermott.